It is typically about the Difference Between Ordinary Water And Heavy Water. It is known condition that 70 percent of the Earth’ surface covered with water which mean higher proportion of the hydrogen isotope makes the clear difference between water and hard water. After exploring the major difference now we move forward towards the normal and deuterium water known as hard water. The normal water molecule means the two hydrogen atoms and one oxygen atom. Deuterium is very much the same as per the normal hydrogen. It contains the normal proportion. Extra neutron added to the weight of the atom that means it is heavier than normal.
Difference Between Ordinary Water And Heavy Water
Table Contents
The water molecule added has so much power indeed. There is lots of difference between the ordinary water physical and chemical properties as compare to hard water. The freezing point of the ordinary water is remain as Zero degrees as compare to the heavy water which has the 3.82 degrees Celsius. Therefore it is normal to say that freezing point of the water is zero degrees Celsius in any aspect. Heavy water also has the higher boiling point when compare to the ordinary water. These elements endorse that page contained the information about the difference between ordinary water and heavy water.
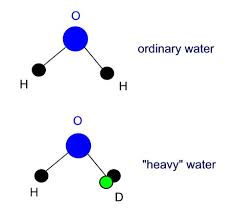
Comparison Between Ordinary Water And Heavy Water
The density does matter here so far that means the heavy water has the higher level of PH and ordinary water has the PH level. The 7.41 when compared to the water’s PH value of 7. In terms of dynamic viscosity, heat fusion and heat of vaporization are some highlighted differences presented here. The below given table ensured the
Difference Between Ordinary Water And Heavy Water
| Ordinary Water | Heavy Water |
| Freezing Point of water is Zero degrees Celsius | Freezing point of hard water is 3.82 degrees Celsius |
| Boiling point of water is 100 degrees | Boiling point of hard water is 101.4 degrees |
| The PH value of water is 7 | The PH value of hard water is 7.41 |
| Viscosity, heat fusion and heat of vaporization of water is lower than heavy water | Heavy water has higher values of Viscosity, heat fusion, heat of vaporization |
| Hydrogen bonds per molecule present in water lower than ordinary water | Hydrogen bonds per molecule present in water higher than ordinary water |
Check Also: Difference Between Altitude And Latitude
While concluding the difference between ordinary water and heavy water, it can say that normal water has the two hydrogen atoms and one oxygen atom. It means the normal molecule made by such aspect. The heavy water is also water but it contains a higher proportion of the hydrogen isotope. Heavy water has a more tetrahedral shape that means the broader structure means as per the designed approach.
